|
12/24/2023 0 Comments Type fu reviews 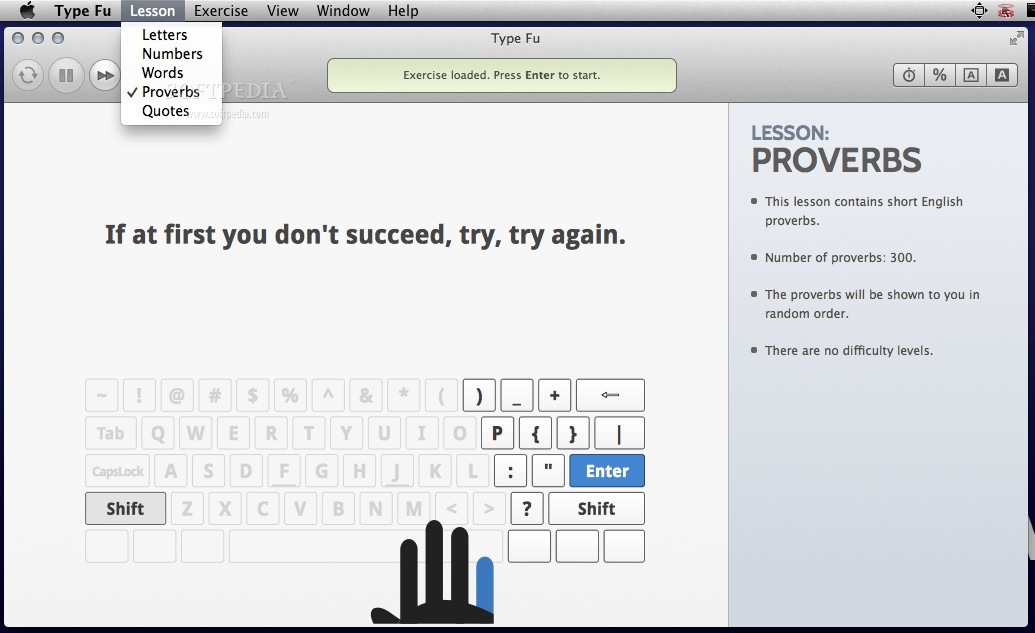
Based on the available evidence, the strength of any recommendations for the use of these 2 agents in the primary treatment of these tumors is weak. Adverse event intensity ranged from mild to severe erythema, pruritus, and pain were common.Įvidence supports the use of topical imiquimod as monotherapy for superficial BCC and topical fluorouracil as monotherapy for superficial BCC and SCC in situ. Up to 100% and 97% of patients applying imiquimod and fluorouracil, respectively, experienced at least 1 adverse event. Fluorouracil use produced the following clearance rates: 90% for superficial BCC and 27% to 85% for SCC in situ. Imiquimod use produced the following clearance rates: 43% to 100% for superficial BCC, 42% to 100% for nodular BCC, 56% to 63% for infiltrative BCC, 73% to 88% for SCC in situ, and 71% for invasive SCC. We calculated the rate of clearance and adverse effects for BCC subtypes and invasive and in situ SCC treated with topical imiquimod or fluorouracil.Ĭlearance rates varied by drug regimen, and most of the studies lacked long-term follow-up. Prospective, retrospective, and case studies in English containing a minimum of 4 subjects and a 6-month follow-up or posttreatment histologic evaluation. MEDLINE, CANCERLIT, and Cochrane databases. 
To conduct a systematic review to determine clearance rates and adverse effects of topical imiquimod or fluorouracil therapy in the treatment of nonmelanoma skin cancers such as basal (BCC) and squamous cell carcinoma (SCC), and to develop recommendations for the use of topical imiquimod or fluorouracil to treat BCC and SCC.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
